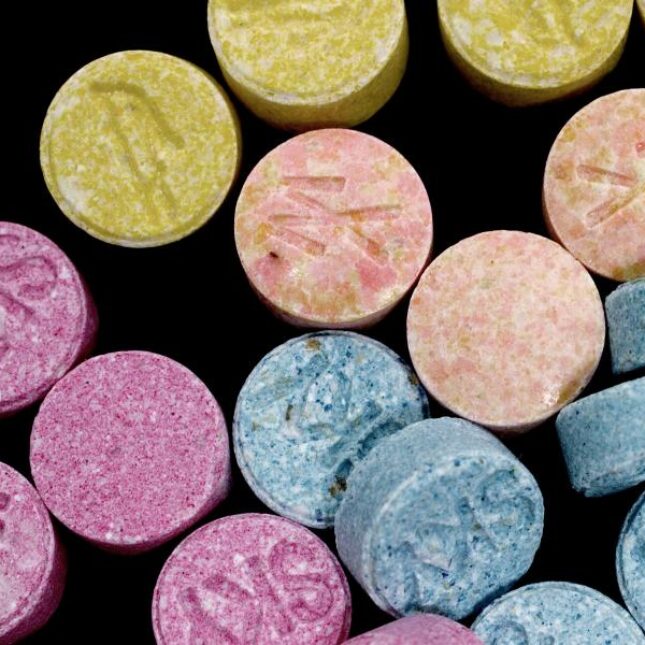
At a heated advisory committee meeting convened by the Food and Drug Administration last week, regulators repeatedly expressed frustration that Lykos, a company seeking approval of MDMA-assisted therapy to treat PTSD, failed to follow instructions and track positive feelings such as “euphoria” that could be used to inform understanding of the drug’s addiction potential.
The missing data, said clinical reviewer David Millis, were “a major concern.”
Lykos executives said they’d simply misunderstood the FDA’s instructions, but three former employees told a different story. The company was long aware it was required to collect such data, they said, and simply failed to do what was needed amid widespread disorganization. Employees described waiting in vain for a protocol to collect the data to be approved, while executives seemed distracted dealing with other problems.
This article is exclusive to STAT+ subscribers
Unlock this article — plus in-depth analysis, newsletters, premium events, and networking platform access.
Already have an account? Log in
Already have an account? Log in
To submit a correction request, please visit our Contact Us page.











STAT encourages you to share your voice. We welcome your commentary, criticism, and expertise on our subscriber-only platform, STAT+ Connect